
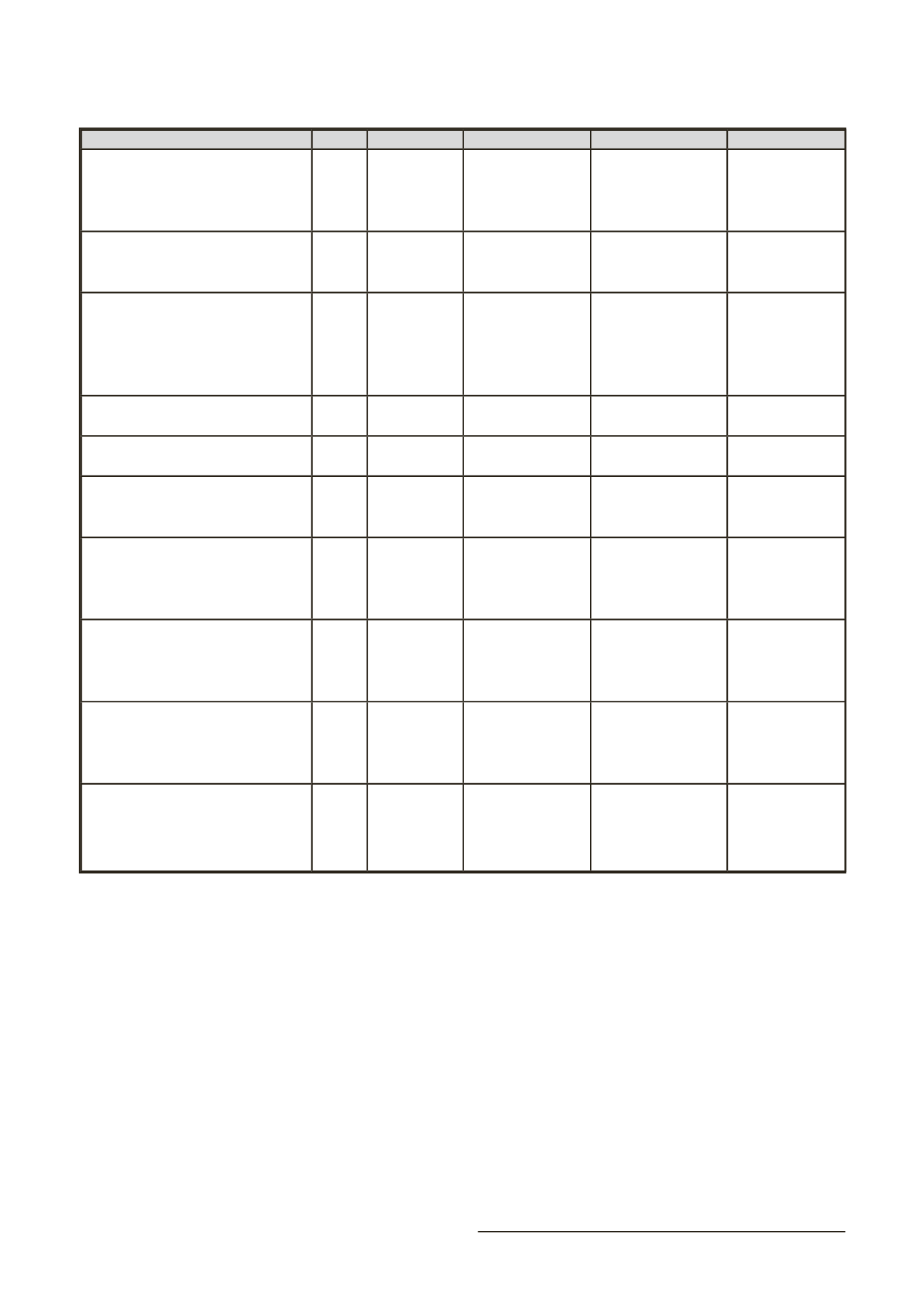
143
Kevin Doello
Nanopartículasmagnéticas y cáncer
Ensayo
Año
Estado
Código
Institución
Fase
Magnetic nanoparticle
thermoablation-retention and
maintenance in the prostate: a Phase 0
study in men
2014 Reclutando
NCT02033447
University College,
Londres
Fase 0
Ferumoxytol - iron oxide nanoparticle
magnetic resonance dynamic contrast
enhanced MRI
2013
Activo
NCT01895829 MD, Anderson Cancer
Center
Fase 0
Pre-operative nodal staging of
thyroid cancer using ultra-small
superparamagnetic iron oxide
magnetic resonance imaging
(USPIO MRI): preliminary study
2013 Reclutando
NCT01927887 Masachussets General
Hospital
No
Proporcionado
USPIO magnetic resonance imaging
(MRI)
2013 Reclutando
NCT01815333 MD, Anderson Cancer
Center
No
Proporcionado
Ferumoxytol-enhanced MRI in adult/
pedi sarcomas
2012 Desconocido NCT01663090
Dana-Farber Cancer
Institute
No
Proporcionado
A novel magnetic needle using iron
oxide nanoparticles for the detection
of leukemia
2011 Completado
NCT01411904 Universidad de Nuevo
México
No
Proporcionado
Pre-operative staging of pancreatic
cancer using superparamagnetic iron
oxide magnetic resonance imaging
(SPIO MRI)
2009 Desconocido NCT00920023 Masachussets General
Hospital
Fase 4
Assessing dynamic magnetic
resonance (MR) imaging in patients
with recurrent high grade glioma
receiving chemotherapy
2008 Finalizado
NCT00769093
Knight Cancer
Institute
Fase 1
A validation study of MR
lymphangiography using SPIO, a new
lymphotropic superparamagnetic
nanoparticle contrast
2005 Terminado
NCT00147238 MD, Anderson Cancer
Center
No
proporcionado
Pelvic lymph node metastases from
bladder and prostate cancer magnetic
resonance imaging with ultrasmall
superparamagnetic iron oxide sinerem
2004 Finalizado 2004-004567-29 Instituto Nacional
para la Investigación
del Cáncer (Italia)
Fase 3
Tabla 2. Principales ensayos clínicos con nanopartículas magnéticas para el diagnóstico y tratamiento del cáncer.
REFERENCIAS BIBLIOGRÁFICAS
1.
Ferlay J, Soerjomataram I, Ervik M, et al. GLOBOCAN 2012 v1.0,
Cancer Incidence andMortalityWorldwide: IARC CancerBase No.
11 [Internet]. Lyon, France: International Agency for Research on
Cancer; 2013 [citado 25 de mayo. 2015]
http://globocan.iarc.fr.
2.
Bray F, Ren J-S, Masuyer E, Ferlay J. Global estimates of cancer
prevalence for 27 sites in the adult population in 2008. Int J
Cancer J Int Cancer. 2013;132:1133-45.
3.
Schmid G. Nanoparticles: From Theory to Application. Wiley-
VCH Verlag GmbH & Co.:Germany; 2010.
4.
Maeda H. Toward a full understanding of the EPR effect in
primary and metastatic tumors as well as issues related to its
heterogeneity. Adv Drug Deliv Rev. 2015.
5.
Akbarzadeh A, Samiei M, Davaran S. Magnetic nanoparticles:
preparation, physical properties, and applications in
biomedicine. Nanoscale Res Lett. 2012;7:144.
6.
Maeda H. Tumor-selective delivery of macromolecular
drugs via the EPR effect: background and future prospects.
Bioconjug Chem. 2010;21:797-802.
7.
Widder KJ, Senyei AE, Ranney DF. Magnetically responsive
microspheres and other carriers for the biophysical
targeting of antitumor agents. Adv Pharmacol Chemother.
1979;16:213-71.
8.
Sun C, Lee JSH, Zhang M. Magnetic nanoparticles in MR
imaging and drug delivery. Adv Drug Deliv Rev. 2008;60:1252-
65.
9.
Wang Y-XJ. Superparamagnetic iron oxide based MRI
contrast agents: Current status of clinical application. Quant
Imaging Med Surg. 2011;1:35-40.

















